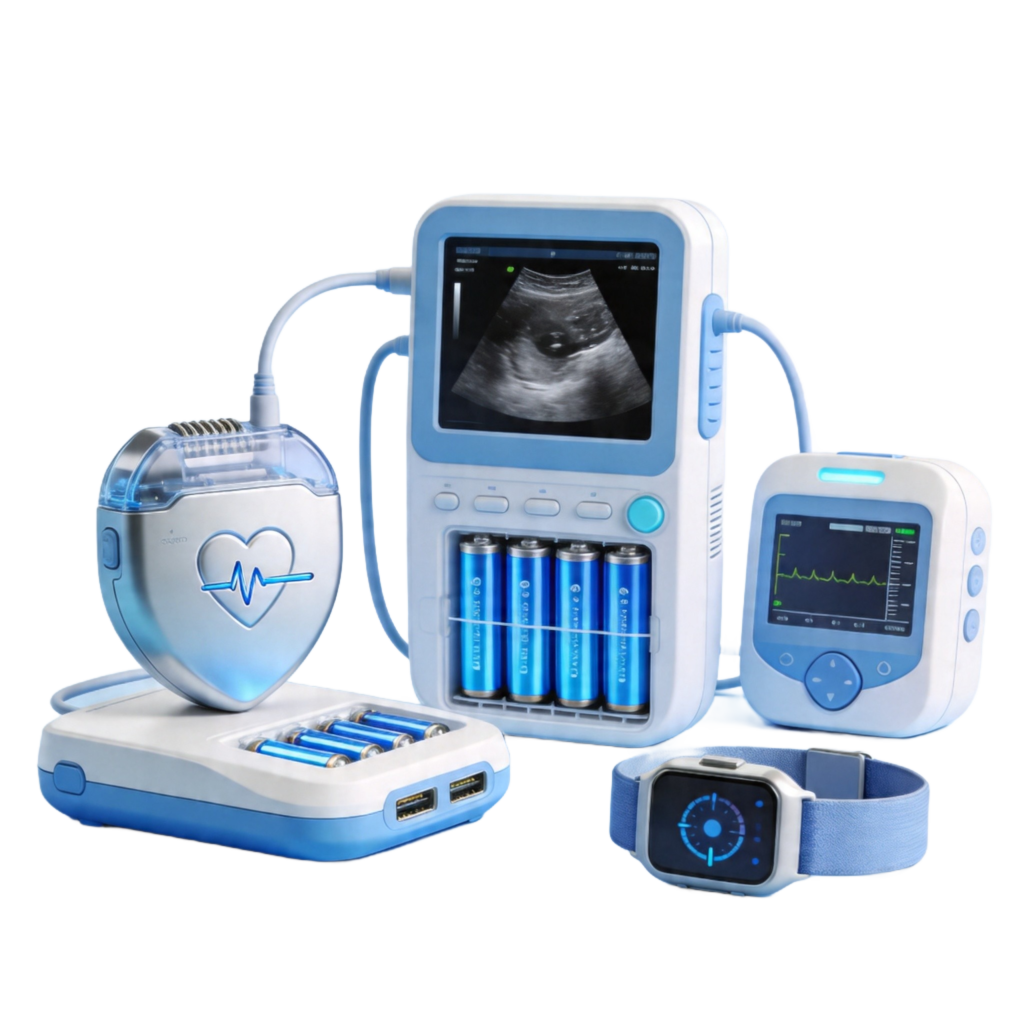
The medical device lithium battery market is experiencing rapid growth. In 2026, the global market size exceeds ¥15 billion, with a compound annual growth rate of 18.7%. Three key factors drive this expansion: portable device trends, aging populations, and continuous technology advancement.
Core Applications of Medical Device Lithium Battery
Implantable Medical Devices
Cardiac pacemakers represent the most demanding application. Lithium-iodine batteries deliver 210-270 Wh/kg energy density, achieving 12 years of operation in devices weighing only 2 grams. Implantable Cardioverter Defibrillators (ICDs) similarly rely on high-density lithium battery technology.
External Link: Medtronic Micra AV Product Information
Portable Diagnostic & Monitoring Equipment
Portable ultrasound devices using polymer lithium batteries reduced weight from 8kg to under 2kg, with 6+ hours of runtime. 62% of global infusion pumps use rechargeable lithium solutions, improving energy efficiency by 36%.
Wearable Health Monitoring Devices
Flexible lithium batteries withstand 100,000+ bending cycles while maintaining 90% capacity, conforming comfortably to skin contours.
Five Critical Selection Criteria for Medical Device Lithium Battery
1. Safety & Certification Standards
Medical device lithium battery must comply with ISO 13485, IEC 60601-1, IEC 60079, and other medical-specific certifications. Intelligent BMS monitors voltage, current, and temperature in real-time, actively disconnecting during anomalies.
External Link: ISO 13485 Medical Device Quality Management
2. Energy Density & Runtime
| Battery Type | Energy Density | Characteristics |
|---|---|---|
| LiFePO4 | 120-160 Wh/kg | Long cycle life, high safety |
| NMC | 200-250 Wh/kg | Ideal for size-sensitive devices |
| Polymer Pouch | Up to 280 Wh/kg | Customizable shapes available |
3. Cycle Life & Reliability
Medical device lithium battery requires ≥80% capacity retention after 500+ cycles. Long-term implantable devices demand 10+ years of stable operation.
4. Extreme Condition Adaptability
Must withstand -40°C low temperature, 130°C sterilization, IP67/IP68 water/dust resistance, and other demanding environments.
5. Customization Capabilities
End-to-end customization—including cell selection, BMS development, structural design, and thermal simulation—represents a manufacturer’s core competitiveness.
Technology Trends
Solid-state batteries have achieved 15% penetration in medical applications in 2026, with energy density exceeding 400 Wh/kg. Intelligent BMS provides 90-day advance failure prediction, reducing device downtime by 70%.
Choosing a medical device lithium battery partner with medical-grade certifications, comprehensive BMS protection, and deep customization capabilities is essential for medical device manufacturers to build competitive products.
